USP-ID
About USP-ID
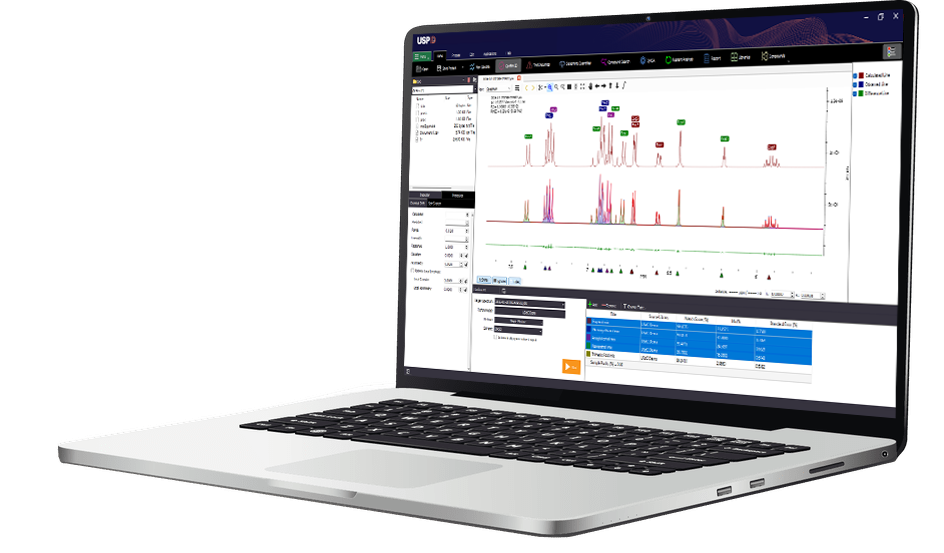
By combining a database of quantum mechanical models with smart algorithms USP-ID software provides an easy-to-use platform for detailed chemical analysis. The platform allows scientists to analyze NMR spectra for identification and quantification of a wide range of chemical components in simple solutions and complex mixtures.
Identification
& Quantitation
Automatic Chemical ID: The “Confirm ID” workspace enables automatic chemical identification by allowing users to screen samples against a broad library of reference compounds. Both targeted workflows, where users select specific chemical references in advance, and non-targeted workflows, where no pre-selection is made, are fully supported.
Find Impurities: The “Find Impurities” feature eliminates manual spectral impurity scans by identifying and labeling signals that do not correspond to a confirmed chemical. Select a signal in the “Find Impurities” workspace to have it highlighted in the spectral viewer for guided spectral review.
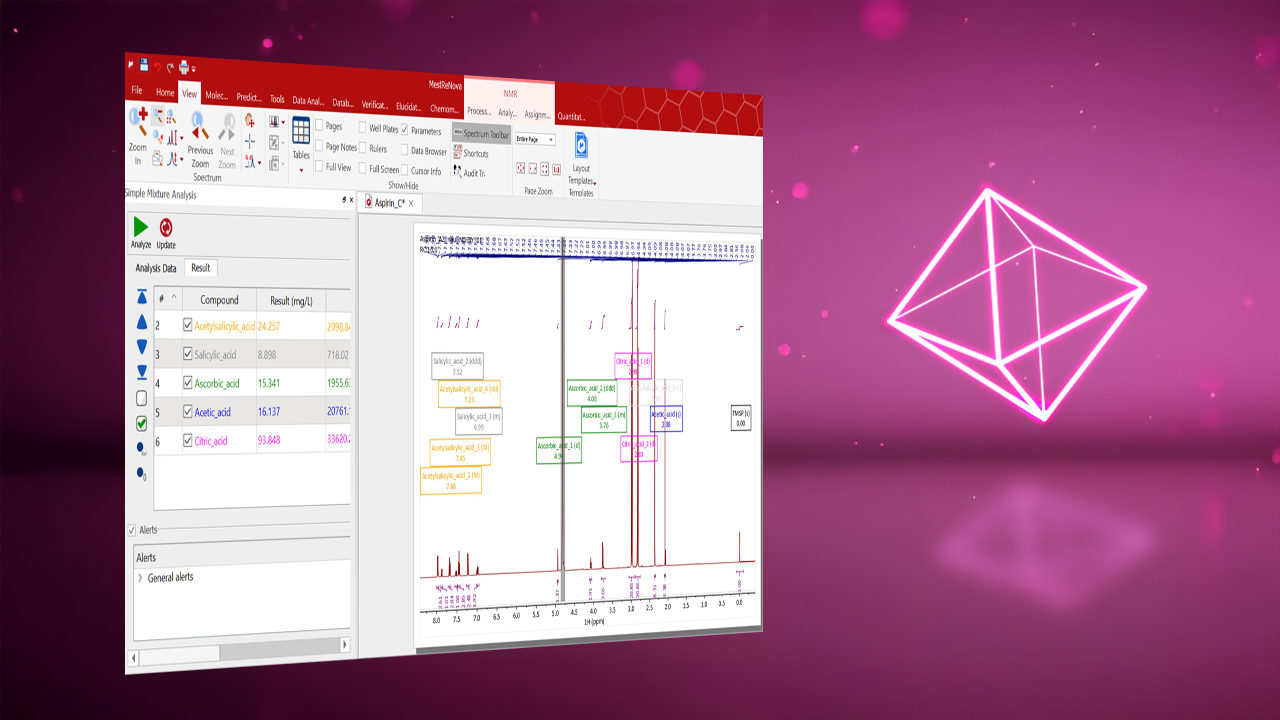
Automatic Quantitation: The “Determine Quantities” feature eliminates time consuming, manual integration of signals and enables fast quantification based on all matched signals by comparison to an internal standard of known concentration.
Report Generator: Generate shareable PDF reports that detail the reproducible analysis steps and results. Reports contain relevant tables of matched compounds and purity values.
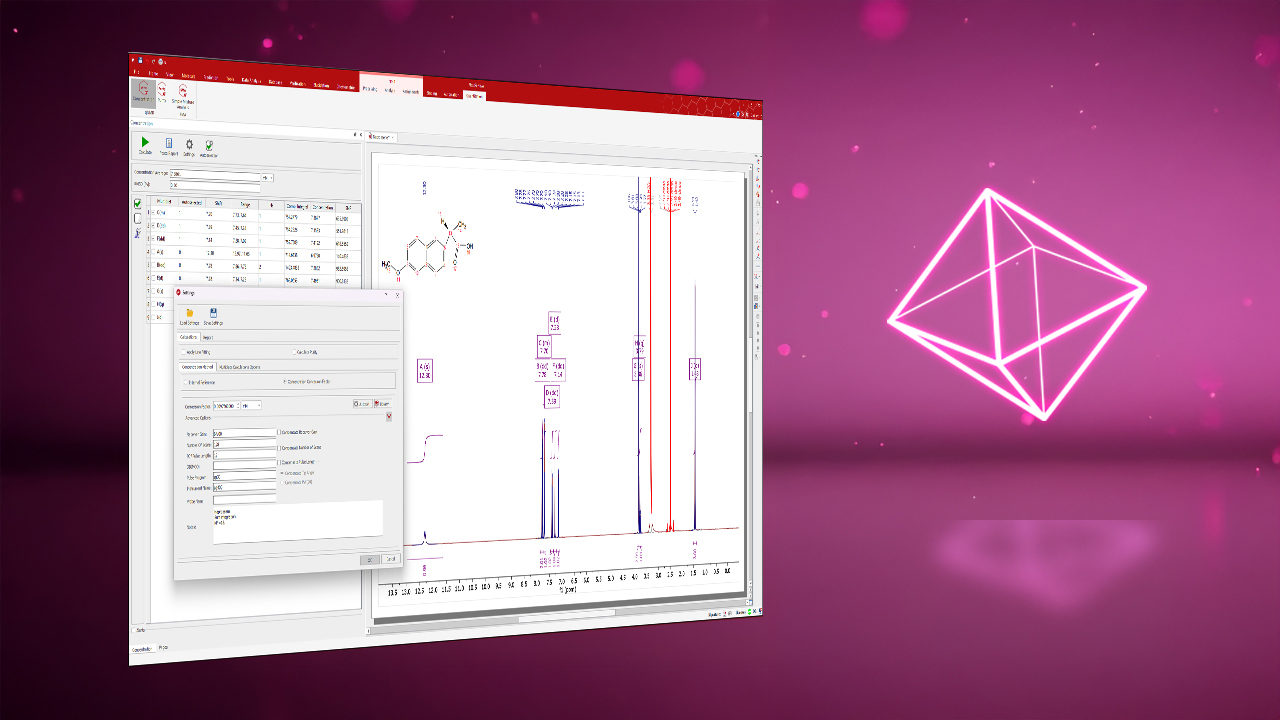
Customizable Libraries: Custom libraries are kept only on the local PC and can be used for the full range of automated NMR data analysis workflows. Drop in new custom libraries or models in the library manager.
Information Workspace: See current analysis results from “Confirm ID”, “Find Impurities”, and “Determine Quantities” in the information workspace found underneath the spectral viewer.
Automatic Peak Labeling: After “Confirm ID” runs, all identified peaks are automatically matched to compounds and labeled in the spectral viewer, making results easier to verify and share.
Multi-library Analysis: Select one or multiple libraries for non-targeted spectral analysis. “Confirm ID” can search multiple libraries simultaneously for matching compounds, including custom libraries kept only on the local PC.
Selectable Analytical Methods: Different analysis methods are available for automatic chemical identification and can be selected from a dropdown in the information workspace. Users can modify and save analysis methods for future use.
Benefits
Easily Identify Chemicals & Impurities: USP-ID automatically identifies, quantifies and labels a broad range of chemical components in complex mixtures. With just 10 minutes of video training NMR becomes more approachable to all.
Resolve Overlapping Peaks: Improve spectral interpretability with quantum-mechanical models based on USP reference standards and smart analytics. Identify and quantify chemicals, even when no single signal is distinct.
Customize Your Databases Locally: Leverage USP-ID automation capabilities with drop-in support for models and databases that users create and own. USP can support with custom model development.
Quantum Mechanical Spectral Analysis is at the core of USP-ID
USP-ID combines libraries of quantum mechanical models with automation algorithms to identify and quantify chemical components of diverse spectra.
Try USP-ID yourself by requesting a demo.
USP-ID can be used with USP’s recently revised and published USP quality standards that facilitate the use of nuclear magnetic resonance (NMR) and quantitative NMR as analytical techniques, including for the control of impurities. These standards incude: USP-NF General Chapters Nuclear Magnetic Resonance Spectroscopy, and Applications of Nuclear Magnetic Resonance Spectroscopy. To further support stakeholder adoption of NMR and qNMR, USP Education offers free training and courses, including “Drug Quality Control Applications for qNMR."
The integration of USP-ID into Mnova does not imply approval, endorsement, or certification by USP of Mestrelab products, nor does it imply that any other brand or product was judged to be unsatisfactory or inadequate. As a standard setting organization, USP avoids providing endorsements that create conflicts of interest that interfere or appear to interfere with its impartiality and objectivity.
Curious about our products or services? Let us provide you with the answers you need. Contact us today and discover how we can cater to your needs effectively.