Mnova StereoFitter
About Mnova StereoFitter
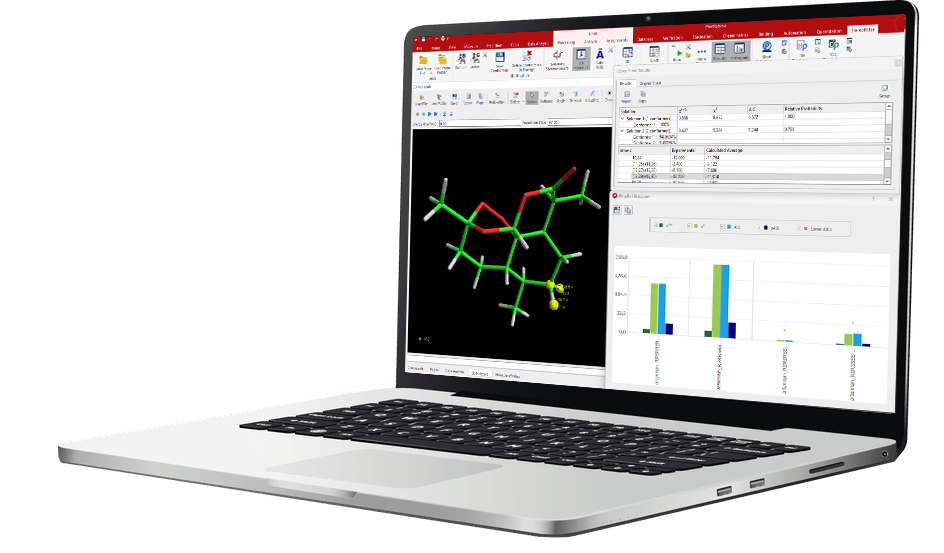
Mnova StereoFitter enables 3D structural and stereochemical analysis by combining experimental NMR observables, including NOEs, RDCs, J-couplings, chemical shifts, and anisotropic parameters, with predicted values to evaluate and rank stereochemical candidates. It generates conformers, computes theoretical NMR data, and fits them to experimental restraints using statistical scoring methods to identify the most probable 3D configuration or conformation.
Analysis
Features
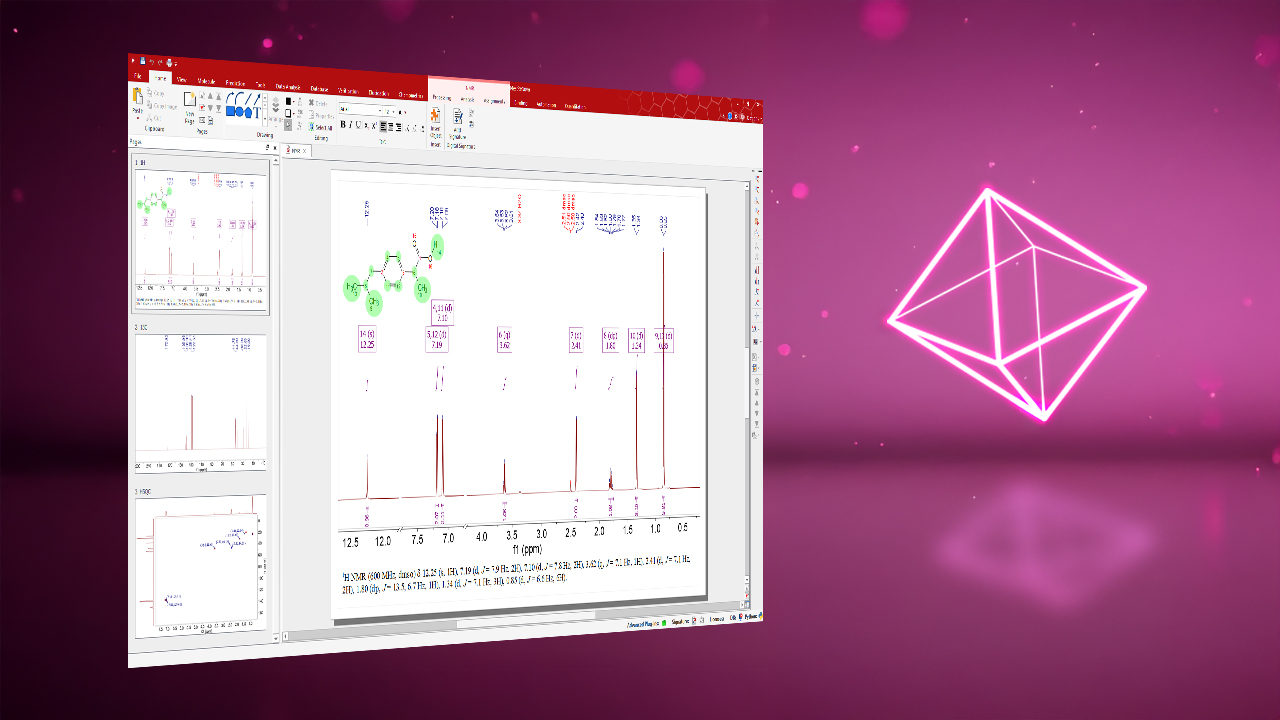
Multi-Technique Input Support
Probabilistic 3D Structural Analysis
Flexible Structure Input
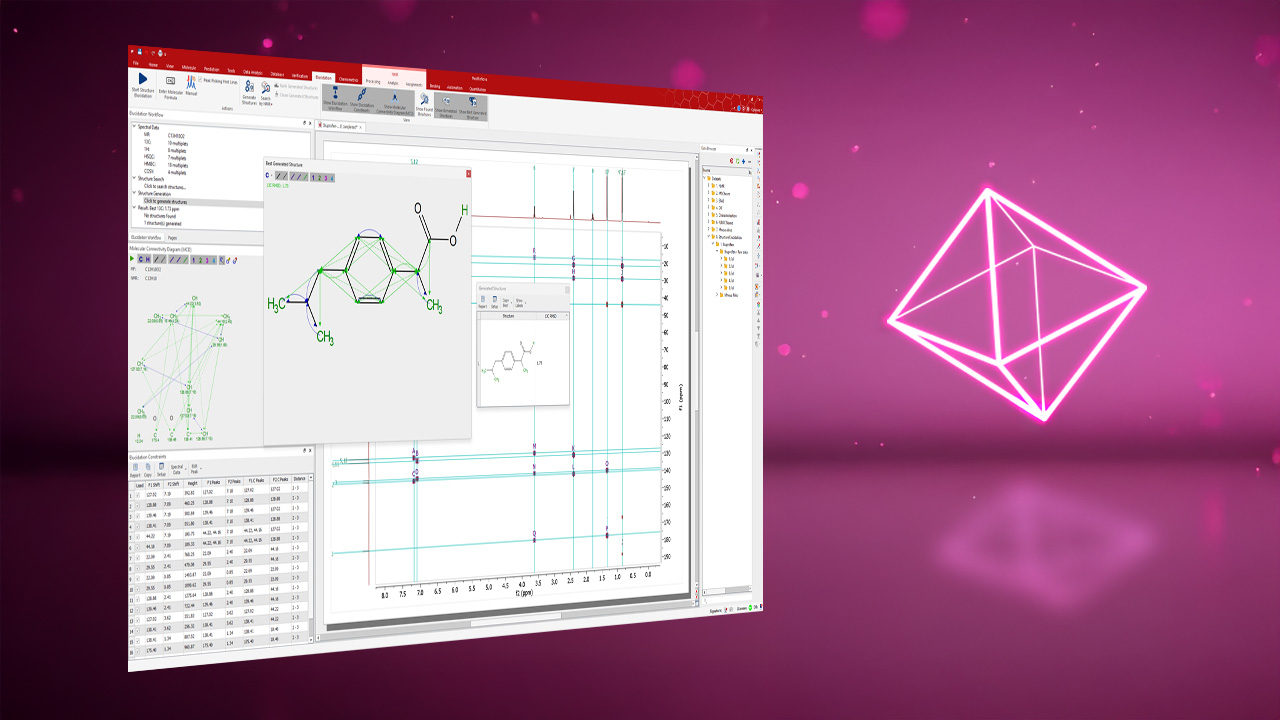
Potential 3D configurations and conformations of a molecule can be generated either within the software or imported from external sources. Compatible file formats include SDF, XYZ, MAE, and outputs from quantum chemistry programs such as Gaussian09/16, Orca, Jaguar, NwChem, ReSpect, and more.
Integrated Visualization Tools
NOE-Based 3D Structure Determination
RDC- and Chemical Shift–Based 3D Structure Determination
DFT-Based Chemical Shift Prediction
Benefits
Rapid Structural Insights: Automating the generation, fitting, and evaluation of stereochemical candidates saves manual effort and significantly speeds structural determination.
Confident 3D Structure Determination: Combining complementary NMR observables (NOEs, RDCs, J-couplings, and chemical shifts) enables confident resolution of challenging stereochemical and conformational problems beyond the reach of single data types.
Time Savings & Efficiency: StereoFitter enables chemists to locally assess plausible structures, ensuring computational hand-offs are focused, informed, and far more efficient.
Clear Results Visualization: Ordered results and interactive 3D models make it easy to present and defend structural conclusions.
Publications
- Liu, Y., Navarro-Vázquez, A., R. Gil, R., Griesinger, C., E. Martin, G., Williamson, R. T. Application of anisotropic NMR parameters to the confirmation of molecular structure. Nature Protocols 14, 217-247 (2019). https://doi.org/10.1038/s41596-018-0091-9
- Navarro-Vázquez, A., R. Gil, R., Blinov, K., Computer-Assisted 3D Structure Elucidation (CASE-3D) of Natural Products Combining Isotropic and Anisotropic NMR Parameters. Journal of Natural Products 81, 203-210 (2018). https://doi.org/10.1021/acs.jnatprod.7b00926
- Liu, Y., Saurí, J., Mevers, E., Peczuh, M. W., Hiemstra, H., Clardy, J., Martin, G. E., Williamson, R. T. Unequivocal determination of complex molecular structures using anisotropic NMR measurements. Science 356, eaam5349(2017). https://doi.org/10.1126/science.aam5349
Curious about our products or services? Let us provide you with the answers you need. Contact us today and discover how we can cater to your needs effectively.