Chrom QC
About Chrom QC
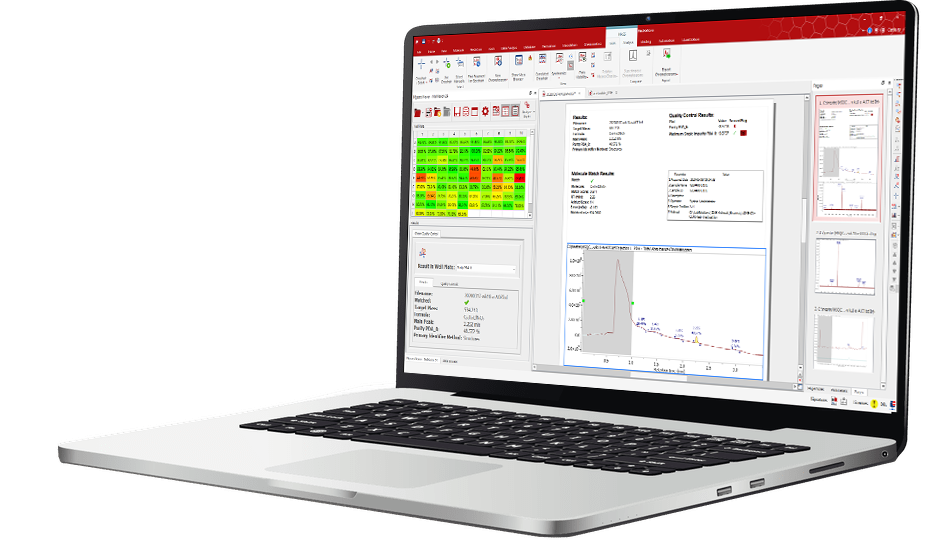
Chrom QC automates the assessment of sample purity and concentration from LC-MS and GC-MS data, eliminating manual data inspection and processing. By combining raw data with expected compound information, it delivers reliable identification, quantification, and quality checks at scale. Standardized reports and intuitive visual tools let scientists quickly determine if samples meet quality standards with confidence.
& Purity
Features
Broad Instrument Compatibility: Chrom QC processes LC and GC-MS data from most major instrument vendors without dependence on proprietary formats.
Smart File Matching: The tool automatically associates raw chromatographic data with provided structures, molecular formulas, or target masses.
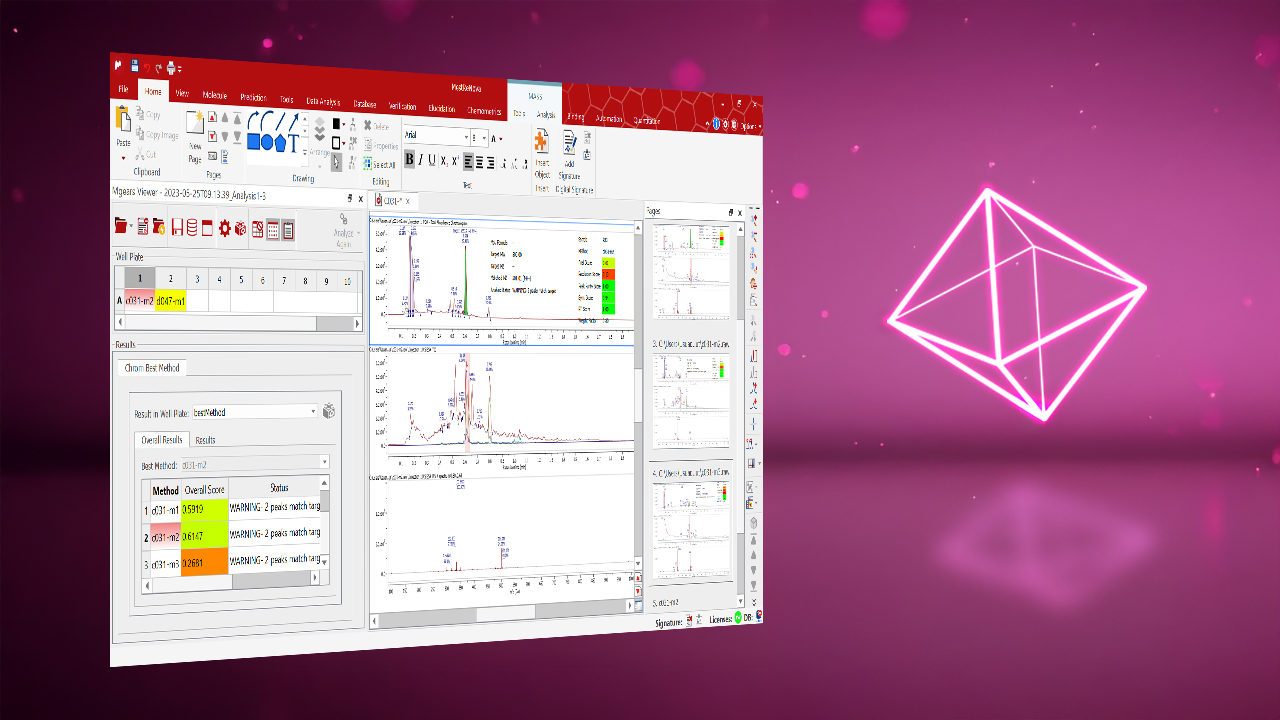
Configurable Data Extraction: Identification of components can be based on structure, molecular formula, mass, or chromatographic peak attributes, and quantification can be easily performed through data gathered from Chrom Cal (additional license needed).
Flexible Peak Identification: Identification and quantification of components can be based on structure, molecular formula, mass, or chromatographic peak attributes.
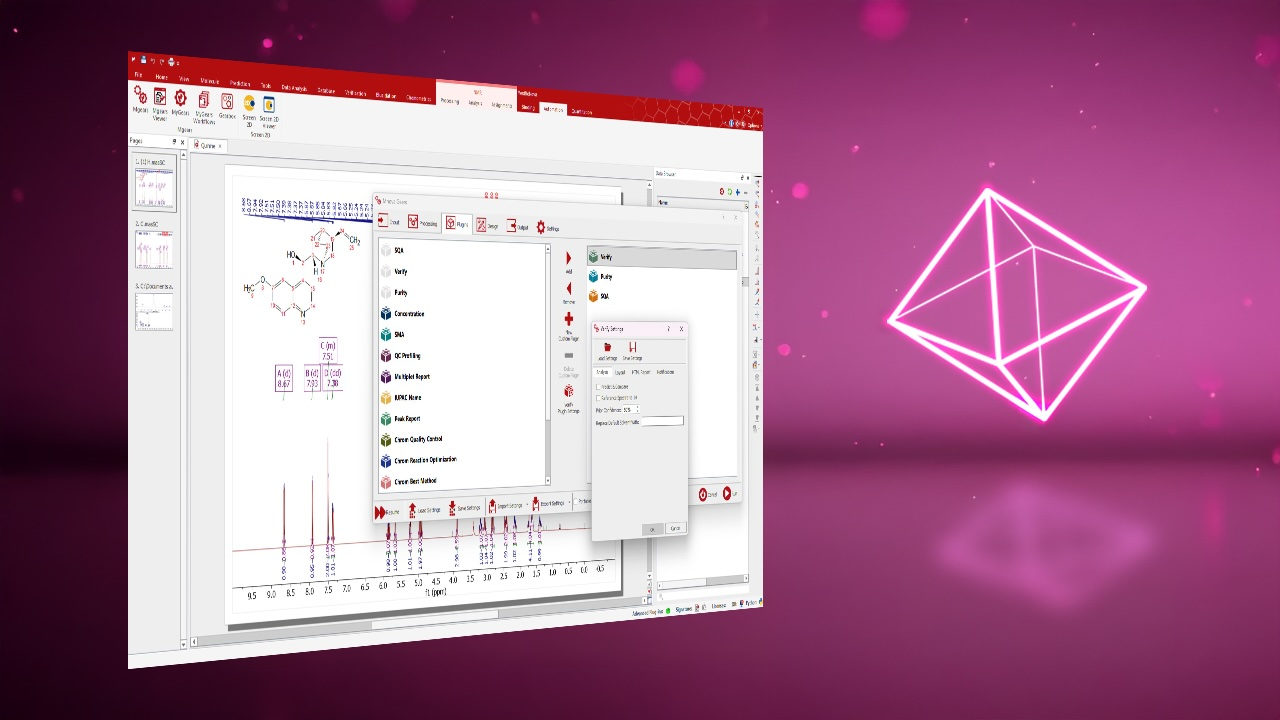
Custom Quality Assessment Rules: Experts can set thresholds for criteria such as purity, concentration, peak shape, retention time, or width, and configure corresponding flags and warning messages.
Customizable Reporting Engine: Branded, templated reports can be generated in multiple formats, including PDF, HTML, CSV, and Mnova files.
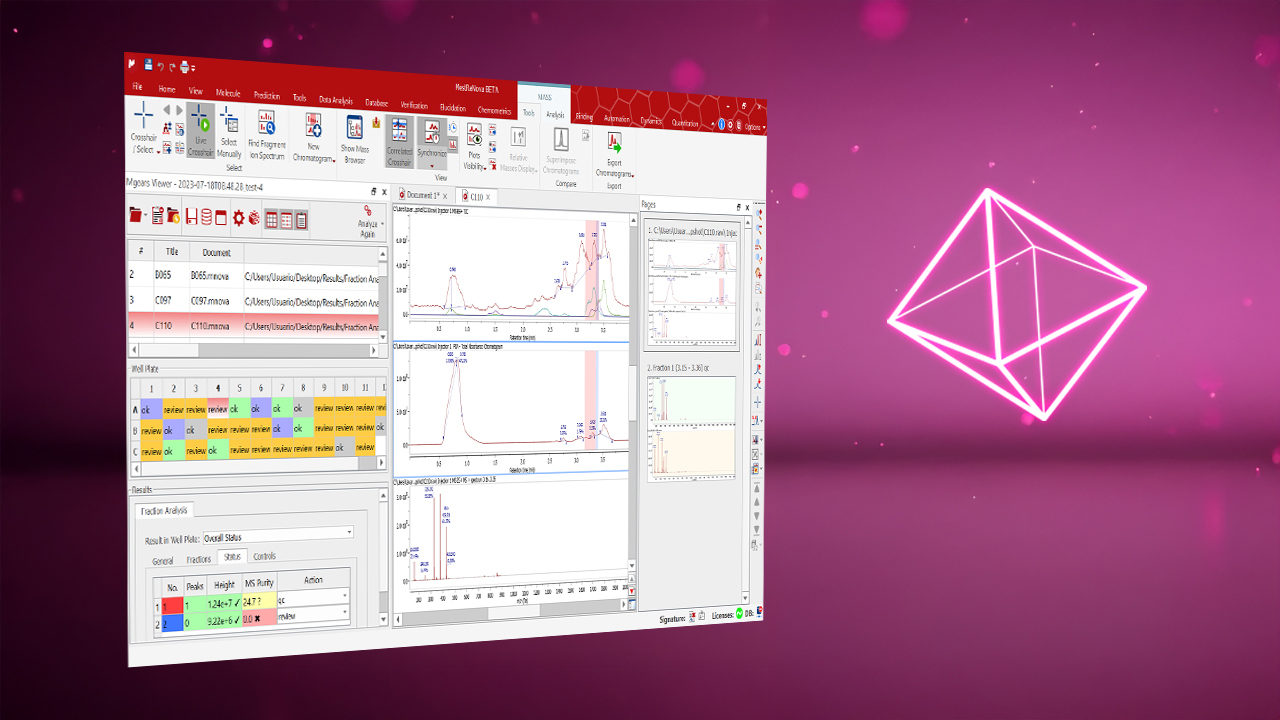
Review-by-Exception Viewer: Out-of-spec results are highlighted to enable focused review of only the questionable samples.
Manual Result Correction: A manual peak selection tool is available to adjust results during final QC review.
Benefits
Faster Decisions: Clear, actionable reports are quickly derived from raw data to support rapid project progression.
Analytical Reliability: Consistent QC rules and thresholds eansure reproducible, defensible outcomes across injections and batches.
Data Confidence: Method standardization and automation deliver consistent results with reduced human error.
Streamlined Workflows: Vendor-format independence, automated input handling, and batch execution allow processing of large numbers of chromatographic datasets from any instrument vendor without creating bottlenecks.
Expert Oversight: Interactive result review allows users to validate results, adjust peaks, rerun analyses, and finalize QC judgments confidently.
All-in-one Mnova Suite: Work seamlessly with other Mnova tools, enabling structure verification and complementary analyses with NMR.
Curious about our products or services? Let us provide you with the answers you need. Contact us today and discover how we can cater to your needs effectively.